Can ALS disease progression be slowed?
University of Illinois at Chicago researchers were awarded $300,000 in seed funding to explore therapeutic targets for disease progression in amyotrophic lateral sclerosis, or ALS.
Fei Song, UIC associate professor of neurology and rehabilitation at the College of Medicine, received the Catalyst award from the Dr. Ralph and Marian Falk Medical Research Trust. The award was created to provide funding to high-risk, high-reward research that could accelerate the treatment progress in diseases that have no known cure.
“ALS has been known for over 150 years but is poorly understood, and there are presently no effective therapeutics to stop its progression,” Song said. “Our research focuses on targeting communication between neuronal cells and non-neuronal cells to slow the disease. Targeting disease progression offers an opportunity to treat all ALS patients, not just those with rare genetic abnormalities, which have so far been the focus of most ALS research.”
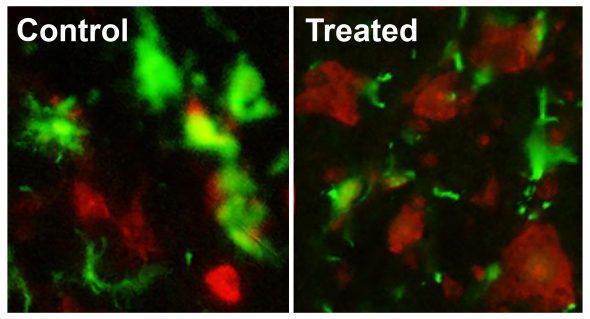
With the new funding, Song will expand on her previous research, which described new human ALS biomarkers and their potential as drug targets. In this research, Song reported that in early-stage ALS, motor neurons — cells that assist muscle movement — in the spinal cord are surrounded by activated cells called microglia. Under normal conditions microglia act as housekeepers and clean up random cellular debris in the central nervous system, but Song’s research suggested microglia also may trigger neuroinflammation and neurodegeneration during early disease progression in ALS patients.
“Our previous studies showed one particular target, a protein called neuregulin that is released from injured motor neurons, activates microglia and neuroinflammatory processes in the central nervous system,” Song said. “In our study, we showed that a neuregulin-blocking drug helped to delay in ALS clinical onset and prolong survival while reducing neuroinflammation and neurodegeneration in mouse models prior to euthanasia.
“This grant will help us further evaluate the therapeutic potential of blocking neuregulin-signaling in a new ALS animal model.”
Categories
Health Sciences Colleges, Research
Topics
ALS, biomarkers, CNS, College of Medicine, grant, molecular biology, neurodegenerative disease
